Peking University, February 1, 2026: A research team led by Prof. Lei Xiaoguang(http://www.chem.pku.edu.cn/leigroup/) at Peking University has achieved a major scientific breakthrough in the field of biosynthesis and biocatalysis. For the first time, they have developed a novel biocatalytic strategy that differs fundamentally from existing methods in terms of mechanism and synthetic logic, enabling transformative synthesis of amide bonds. This seminal discovery was published in
Science on January 29, 2026, under the title: "Engineered aldehyde dehydrogenases for amide bond formation."


The Silent Skeleton of Modern Medicine
Amide bonds are the "bread and butter" of medicinal chemistry. These molecular linkages form the structural backbone of some of the world's most successful drugs, including the blockbuster anticoagulant Apixaban and the leukemia treatment Imatinib. Despite their ubiquity, creating these bonds in a laboratory is a notoriously "messy" affair.
For decades, the pharmaceutical industry has relied on "incumbent but expensive heavyweights"—expensive coupling reagents like DCC; toxic and costly transition metal catalysts like ruthenium or copper. Traditional synthesis is a brute-force process, requiring high-activation-state precursors from acids and wasteful coupling reagents to force the reaction. Now, Prof. Lei Xiaoguang's research group at Peking University has pioneered a biocatalytic shift, "reprogramming" one of nature's most common tools to unseat these industrial heavyweights.

video: Simple and efficient real reaction process
Takeaway 1: Turning Acid-Makers into Amide-Builders
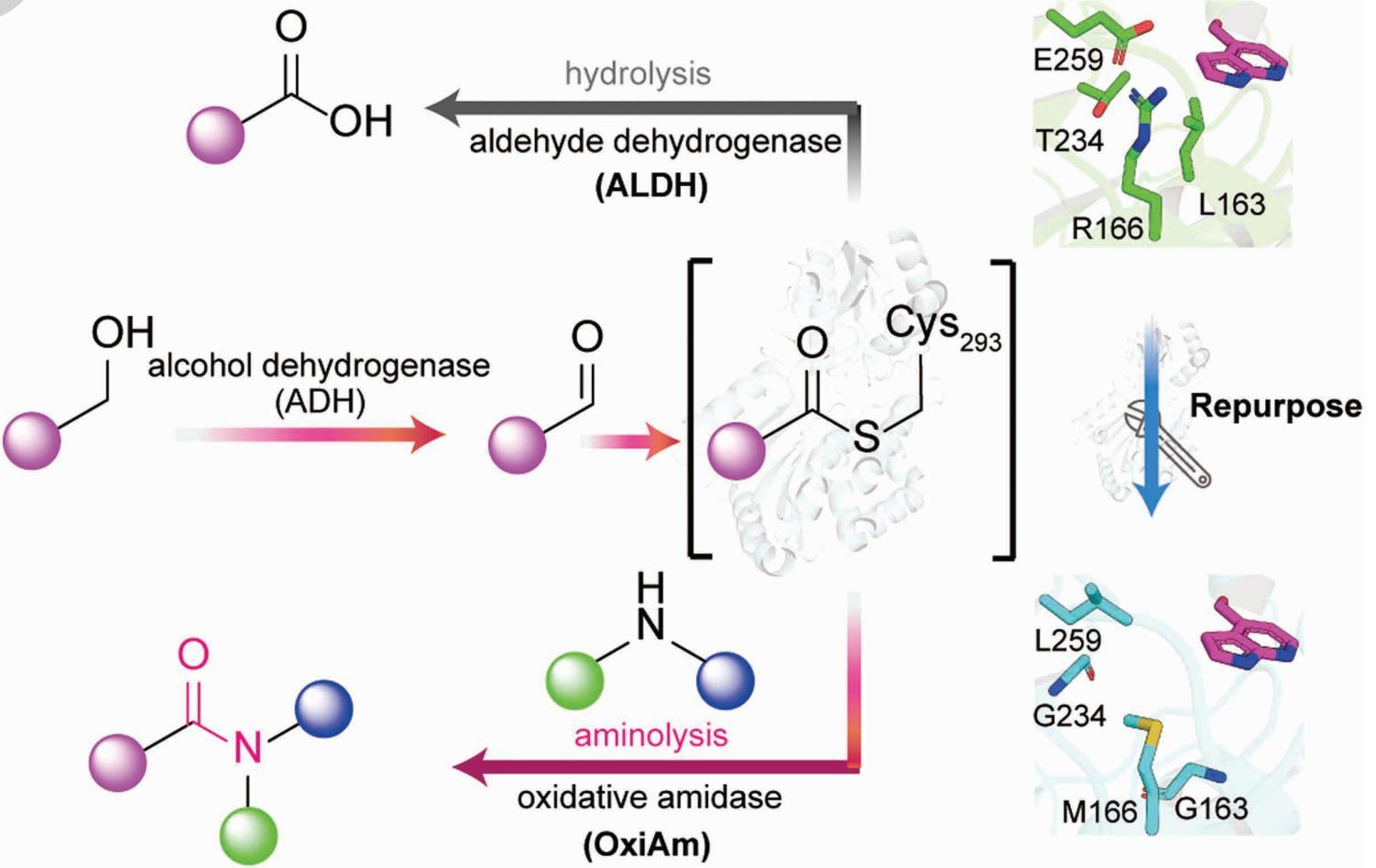
The core breakthrough involves the molecular hijacking of Aldehyde Dehydrogenases (ALDH), a massive superfamily of enzymes found throughout the natural world. Specifically, the team focused on PHBDD (p-hydroxybenzaldehyde dehydrogenase) from the bacterium Pseudomonas putida. Normally, these enzymes perform a simple, essential task: they turn aldehydes into carboxylic acids using water.
The researchers transformed these "acid-makers" into Oxidative Amidases (OxiAms). Mechanistically, the enzyme traps the aldehyde in a high-energy thioester intermediate. In nature, water typically attacks this intermediate to form acid. However, by pulling a "pH lever" to 10–10.5, the scientists ensured that ammonia became nucleophilic enough to outcompete water, hijacking the intermediate to form an amide bond instead.
"The development of a biocatalytic approach for directly forming amide bonds from aldehydes or alcohols will... expand the biocatalytic repertoire and offer advantages for the sustainable synthesis of complex bioactive molecules."
 Takeaway 2: The Hydrophobic Secret—Redesigning the Molecular Pocket
Takeaway 2: The Hydrophobic Secret—Redesigning the Molecular Pocket
Transforming PHBDD required precision "rational design" to overhaul its internal architecture. The team identified two well-conserved hydrophilic residues, E259 and R166, which typically invite water into the catalytic site. In a counter-intuitive move, scientists replaced these with hydrophobic residues, such as Methionine and Leucine.
This swap did more than just "make room"; it changed the electronic preference of the site. Because alkylamines are more hydrophobic and possess higher nucleophilicity than water, they can now outcompete water molecules to attack the thioester intermediate. Additionally, the researchers identified a "gate" at the intersection of the binding pocket and the solvent. By introducing the T234G mutation, they widened this door from 6.1 to 8.1 Å, allowing bulkier amines to enter.
Crucially, this wasn't just a single-enzyme trick. Because these residues are relatively conserved, the researchers established a general principle and successfully applied this "blueprint" to other members of the ALDH family, including ALDH1 and ALDH2.
Takeaway 3: The Alcohol-to-Amide Shortcut
The team extended their logic to create a "one-pot" enzymatic cascade that starts from broadly available aliphatic alcohols. This chemoenzymatic strategy is exceptionally "atom-economical," streamlining the synthesis of high-value compounds like the PARP1 inhibitor Saruparib.
The system integrates three essential components:
• XylB: An alcohol dehydrogenase that converts the starting alcohol into an aldehyde.
• OxiAm: The engineered enzyme that captures that aldehyde to form the amide.
• TpNOX: A recycling enzyme that keeps the system's power source (NADP+) running.
This recycling is fueled by dioxygen (O₂) as the ultimate electron acceptor, making the process incredibly green—leaving behind only benign species rather than toxic metal waste.
Takeaway 4: From Theory to Pharmacy—Real-World Impact
The true test of OxiAms lay in their ability to manufacture modern pharmaceuticals. The Lei group used these biological nano-factories to redesign synthetic routes for several major drugs:
• Vadadustat: An anemia drug traditionally requiring a complex five-step synthesis; OxiAms simplified the process using a de novo designed route.
• Lazabemide: A monoamine oxidase inhibitor produced at gram-scale with a 60% yield.
• Imatinib: To synthesize this leukemia treatment with high precision, the team developed a specific variant—M3-P156V-L259M—which was essential for maximizing selectivity and minimizing the byproducts that typically hinder the reaction.
These results were achieved using nothing more than engineered E. coli cells as the catalyst factory, reaching up to 90% conversion without any expensive transition metals.
"Our findings highlight the potential of oxidative amidases in advancing the synthesis of structurally diverse drug molecules through efficient amide bond formation."
Conclusion: The Future of Sustainable Synthesis
The development of OxiAms marks a pivot in pharmaceutical manufacturing. By shifting from high-activation precursors to "low-activation" compounds like alcohols and aldehydes, we can create high-value drugs with minimal environmental footprints.
These "highly tunable" enzymes prove that the ALDH superfamily is a goldmine for green chemistry. As we continue to map these molecular machines, it raises a compelling question: If we can reprogram one common enzyme to perform the "impossible," which other chemical bottlenecks might nature eventually solve for us? Prof. Lei's group is currently pursuing this direction to creat more new enzymes for efficient, scalable, green and sustainable synthesis of high-value drug molecules.

video:Introduction of Research Results
Read more: https://www.science.org/doi/10.1126/science.adw3365
Edited by: Chen Shizhuo
Source: College of Chemistry and Molecular Engineering




